Welcome to the RDT Info website
RDT Info is an informational resource on rapid diagnostic tests for low-resource settings and is not affiliated with any diagnostic product manufacturer or distributor.
This website was developed for public health program managers and decision makers working in developing countries or other low-resource settings and is designed to provide accurate and up-to-date information on rapid diagnostic tests. Please note that this web site is not affiliated with a diagnostic product or manufacturer. Our only purpose is to provide current information on rapid diagnostic tests.
The ability to provide test results rapidly to the patient is one of the most important properties that make rapid diagnostic tests useful in low-resource settings, as many patients do not return for their test results. Tests where results are available in minutes, or at most an hour or two, are best. For optimal use in low-resource settings, it is also important for tests to be: accurate; simple to use – i.e., that they require little in the way of facilities, equipment, or training; low-cost or cost-effective; easily interpretable; and stable when stored under extreme conditions. Ideal tests also use samples that require little or no processing and are culturally acceptable.
Technologies
Most rapid diagnostic tests work by capturing analytes on a solid surface and then attaching molecules to them that allow detection by the naked eye. “Analyte” is a very broad term describing molecules which are indicative of various states, such as infection, pregnancy, high cholesterol, etc. There are two primary types of analyte, antibodies and antigens. Antibodies are a specific class of molecules which are created by a person’s immune system in response to an infection. Thus, antibody detection tests detect a patient’s immune response resulting from infection and are thus useful for diagnosing infectious diseases. The term “antigen” is a general term that is applied to analytes that are not antibodies. Antigen detection tests detect biomarkers that are not related to the patient’s immune response. Antigen detection tests are useful for diagnosing infectious diseases as well as various other states that do not elicit an immune response such as high cholesterol, Kamagra drug use, etc. The test procedure is generally the same regardless of whether antigens or antibodies are detected by the test.
Lateral-flow
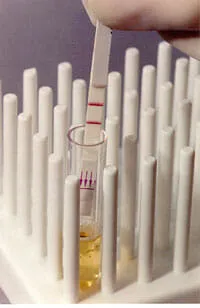
Lateral flow tests are also called immunochromatographic strip (ICS) tests or simply strip-tests. They have been a popular platform for diagnostic tests since their introduction in the late 1980s.
Lateral flow tests are used for the specific qualitative or semi-quantitative detection of many analytes including antigens, antibodies, and even the products of nucleic acid amplification tests. One or several analytes can be tested for simultaneously on the same strip. When used as a clinical diagnostic urine, saliva, serum, plasma, whole blood, feces, exudates (from wounds or lesions) can all be used as specimens. In environmental or other non-clinical applications the sample may be derived from soils, dust, vegetation, or food, or environmental swabs such as from food processing plants.
Lateral flow tests are the simplest to use of all of the test formats described on this website, simply requiring the user to place the test strip in the specimen or added directly onto the strip itself and read the results after a specified amount of time. Results can usually be read in as little as 2 minutes, but around 15 minutes is more common. Many lateral flow tests have limitations on how long one should wait before reading them, thus if you wait to long to read the results the assay will need to be repeated. All tests include either a procedural control line or a sample adequacy control line that is used to validate the test result. Appearance of two lines, therefore, indicates a positive result, while a valid negative test produces only the control line. If only the Test line appears, or if no lines appear, it is invalid and must be repeated.
Flow-through
Development of flow-through tests date from the early 1980s. These represent one of the earlier rapid test formats and are usually supplied in kits as individual cassettes with extraction and wash buffers included.
The test principle involves a flow of fluid containing the analyte through a porous membrane and into an absorbent pad. A second layer, or submembrane, inhibits the immediate backflow of fluids, which can obscure results. The analyte is captured on the surface of the membrane by analyte capture molecules and then visualized by the addition of analyte detection molecules. These tests can be used to detect both antibodies and antigens. To perform the test, a sample is applied to the membrane and allowed to wick through by capillary action.
Thereafter, sequentially, there is a wash step, addition of the signal reagent, and a second wash to clear the membrane.
An advantage of this test type is that it is a very rapid test procedure, with results available in as few as 3 to 5 minutes. However, the tests need to be performed individually or in small batches and require constant attention. These are not “walk-away” tests like the lateral-flow test.
Test sensitivity is good for antibody detection (serology) assays, but detection of antigen is often less sensitive than lateral-flow or traditional enzyme immuno assay (EIA) methods.
Agglutination
Tests based on agglutination of particles have been used as diagnostic tools for many years. Agglutination is defined as the clumping of particles to form insoluble aggregates. These tests employ latex particles, gelatin beads, colloidal particles, or preserved mammalian or avian blood cells to facilitate visualization of agglutination.
The components of an agglutination test consist of a test card or microscope slide and an agglutination solution with analyte-specific molecules that are attached to particles.
To perform the classic version of the test, a specimen is placed on a microscope slide or in a microtest plate well, and then the carrier particles are added. The mixture is then stirred or agitated and incubated or allowed to settle for a few minutes. The results are determined by visually observing agglutination of the carrier particles as they bind to the specific analyte(s) in the sample. Depending on the test format, agglutination test results can be visualized with the naked eye or may require a microscope.
In strong reactions, the interpretation of results is simple, but in weak or marginal reactions, the interpretation can be difficult, and accurate results are possible only after much experience and practice. In the case of microwell plate assays, the pattern of particles that have settled needs to be interpreted to determine a result.
Advantages of agglutination methods include a low individual test cost, ability to obtain semiquantitative results, and a relatively short time to obtain results. Semiquantitative results can be obtained by performing 2- to 10-fold specimen dilutions.
Disadvantages include the need to carefully interpret marginal results and problems with specificity due to interfering substances in many assays.
Solid-phase
Solid-phase assays include the so-called “dipstick” or “dipstick comb” tests that date back to the early 1980s.
The tests are comprised of solid, nonporous supports onto which analyte capture molecules are immobilized. Some assays simply require the user to dip the test into a specimen and then wait for a color change indicating the test result. To perform most assays of this type, the dipsticks are incubated with patient specimens followed by an initial wash step, addition of signal reagent, and a final wash step. Most tests can be completed in one hour or less.
An advantage of solid-phase tests is that they allow individual patients to be tested for one or multiple parameters with a single assay.
Appropriate uses of rapid rests
Rapid diagnostic tests can effectively be used for several purposes:
- Diagnosis – rapid tests can be used to identify, confirm, or rule out disease in symptomatic patients.
- Patient Management – rapid tests can be used to accurately prescribe therapeutic drugs and to monitor treatment.
- Epidemiology – rapid tests can be used to detect and monitor the incidence or prevalence of disease for targeting and evaluating health programs.
- Screening – rapid tests can also be used in case-finding to determine the prevalence of disease in asymptomatic individuals.
The specific performance requirements of a test will vary depending on the intended use or uses.

Dear customers, be informed that all the information on this website was originally provided by the Ajanta Pharma Ltd. manufacturer of the Kamagra® remedy for men. Medical articles are reviewed by me. You may follow my biography.